All substances are made up of tiny particles called atoms.
There are over 100 different types of elements, each with its own sort of atom.

Each element has its own unique name and unique symbol.
For example:
The element copper has the symbol Cu.
The element sulfur has the symbol S.
Remember that:
1. All chemical symbols begin with a capital letter.
2. If an element has a symbol with a second letter, the second letter is always a lower case letter not a capital letter e.g. the symbol for calcium is Ca not CA.
3. Some elements have a symbol based on the element's original Latin or Greek name. E.g. the element called iron has the symbol Fe, based on iron's original Latin name, Ferrum.
Top Tips:
To help scientists remember the names and properties of the various elements, scientists use a special chart called the Periodic Table.
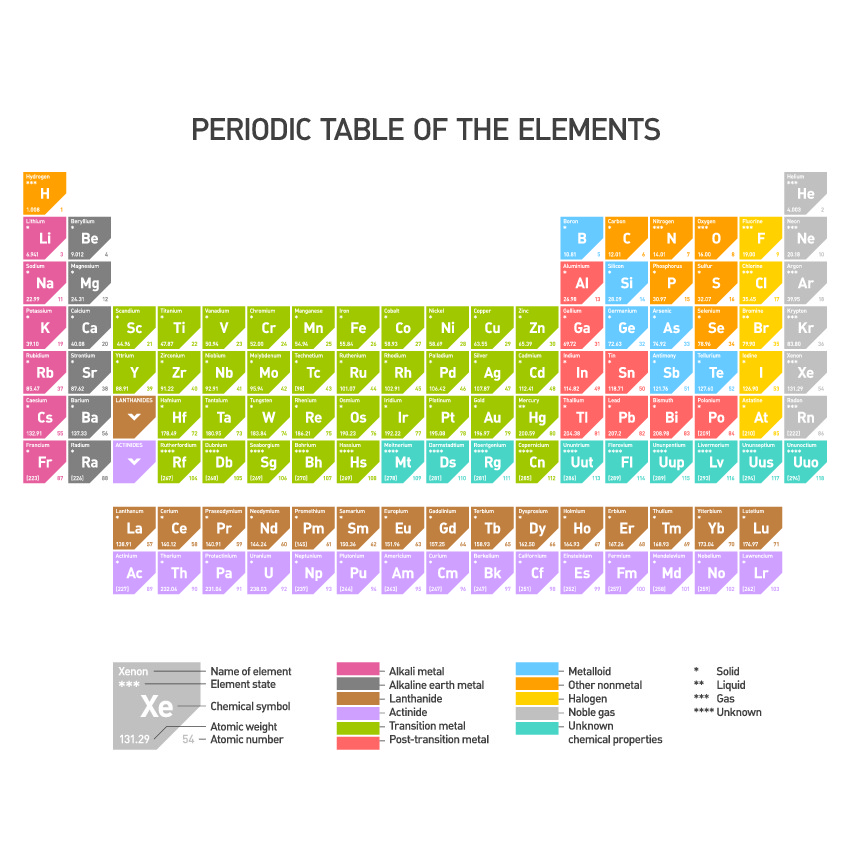
At GCSE level all students are expected to be familiar with the names and symbols of at least the first 20 elements in the Periodic Table. It's very important to be able to recognise the names and the symbols, so the more we use the Periodic Table the more familiar we will be!
Once you have looked at the names and the symbols, practise using them and they'll become second nature to you!
In this activity, students will revise the names and symbols of the first ten elements in the Periodic Table.